 3-D printing has been around for a while. Originally called “rapid prototyping”, stereolithography (SLA) was initially developed in 1980 by the Japanese. The concept never gained momentum until a few years later when American Chuck Hull invented the SLA machine, or 3D printer. This was huge for rapid prototyping of industrial products. However this never made mainstream headlines since machines were costly and most objects warped as the materials hardened.
3-D printing has been around for a while. Originally called “rapid prototyping”, stereolithography (SLA) was initially developed in 1980 by the Japanese. The concept never gained momentum until a few years later when American Chuck Hull invented the SLA machine, or 3D printer. This was huge for rapid prototyping of industrial products. However this never made mainstream headlines since machines were costly and most objects warped as the materials hardened.
3D printers work much like a regular office or desktop inkjet printer. First, a computerized file is created and uploaded to the printer. In an inkjet, the “PRINT” button is depressed and ink is spewed on paper. The difference with a 3D printer is the materials and extra print-cycles. Filaments, instead of ink, are used to create the copy, which could be metal, glass, wood or even human tissue. And to create 3 dimensions, many print cycles layer the filaments.
 In medicine, 3D printing became innovative in 2010, when the first ever 3D printed human bladder was developed out of patient tissue. This could be implanted with lower risk than a standard organ transplant, since an immune system would not reject its own cells.
In medicine, 3D printing became innovative in 2010, when the first ever 3D printed human bladder was developed out of patient tissue. This could be implanted with lower risk than a standard organ transplant, since an immune system would not reject its own cells.
In neurosurgery, 3D printing of complex brain neuroanatomy or spine models can help train residents or help prepare a practicing surgeon for complex surgery. Novel 3D reconstructions help visualize structures in anticipation.
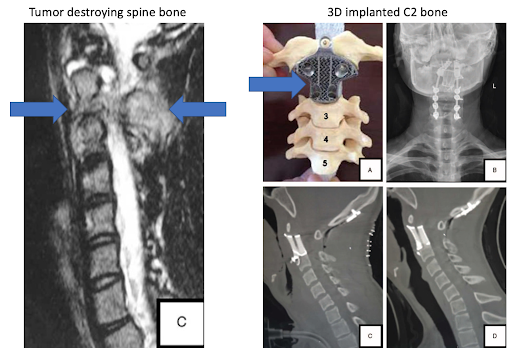
In spine surgery, 3D printed vertebral prostheses with viable structural augmentation properties have been implanted.
More commonly, though, a scoliotic spine can be constructed preoperatively to plan screw trajectories and correction.
Recently, an Israeli group has used 3D printing to individualize neuro-oncology for glioblastoma multiforme (GBM) treatment. While a patient’s
tumor can be reconstructed in relation to the surrounding brain structures for operative planning, 3D printing may also allow oncologists to test different regimens prior to actual postsurgical chemotherapy. After all, it is estimated that about 30-40% of cancer patients are treated with ineffective drugs due to the inability to adequately individualize treatment.
 Scientific breakthrough in the battle against cancer: First 3D printing of glioblastoma cancer tumor
Scientific breakthrough in the battle against cancer: First 3D printing of glioblastoma cancer tumor
After a tumor is surgically removed, the tissue is pathologically diagnosed. Following standard surgery, usually about 30 days of recovery are required prior to starting potential chemotherapy, to allow healing of the incision.
This research group was able to use human and mouse tumor cells to 3D – bioprint a copy of their GBM. Tissue could also reconstruct a genetically similar surrounding brain cells and vasculature. Using the model, the subject’s own blood could be pumped through the 3D tumor model, along with various different chemotherapy regimens. In a few weeks, amidst surgical recovery, the researchers could identify the best possible chemotherapy regimen for the specific tumor.

This 3D printed model could be manufactured on-demand to serve as a drug screening array evaluating chemotherapy response, customized individually to each patient. This may revolutionize theway we treat patients by mimicking their brain tissue microenvironment and predicting specific treatment. The greatest limitation now, is developing more chemotherapy regimens to run through the model.
References
Neufeld L et al. Microengineered perfusable 3D- bioprinted glioblastoma model for in vivo mimicry of tumor microenvironment. Science Advances. 2021.






